Presentation Shows MammaPrint Can Accurately Stratify Recurrence Risk up to 25 Years
IRVINE, CA and AMSTERDAM, March 26, 2014 – Four scientific presentations at the 9th European Breast Cancer Conference (EBCC) underscored the international scope of positive research findings regarding MammaPrint® and other genomic tests in Agendia’s Symphony suite of breast cancer tests.
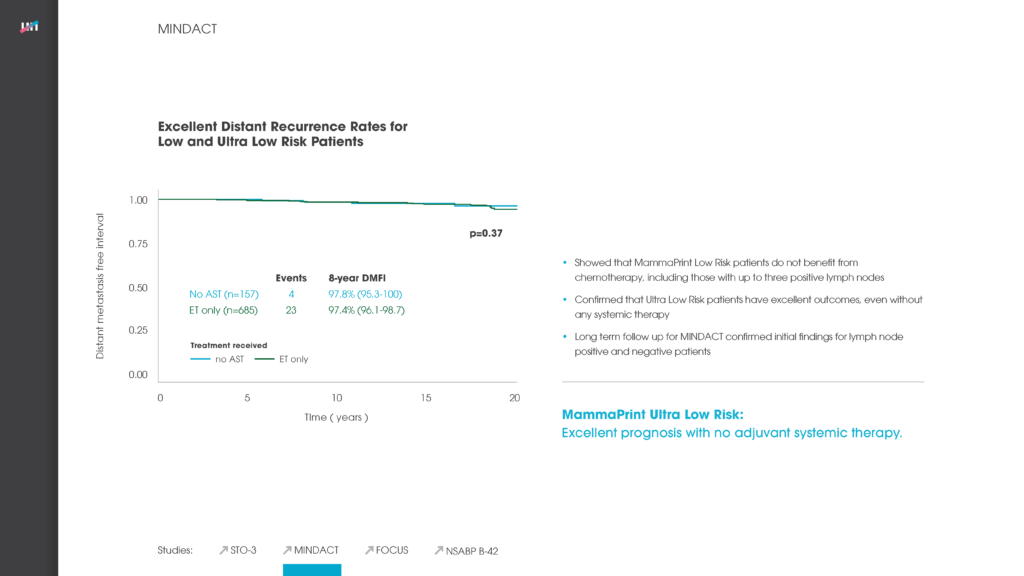
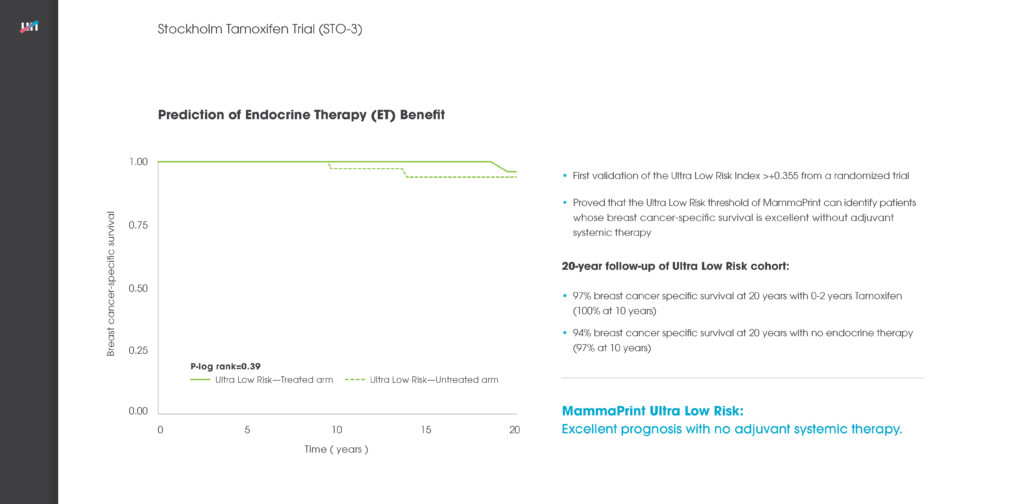
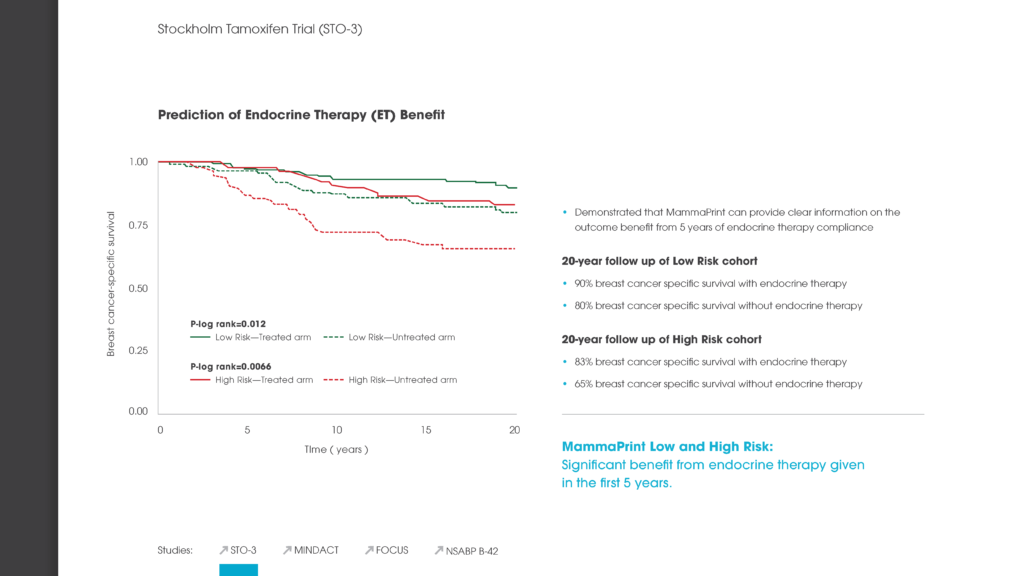
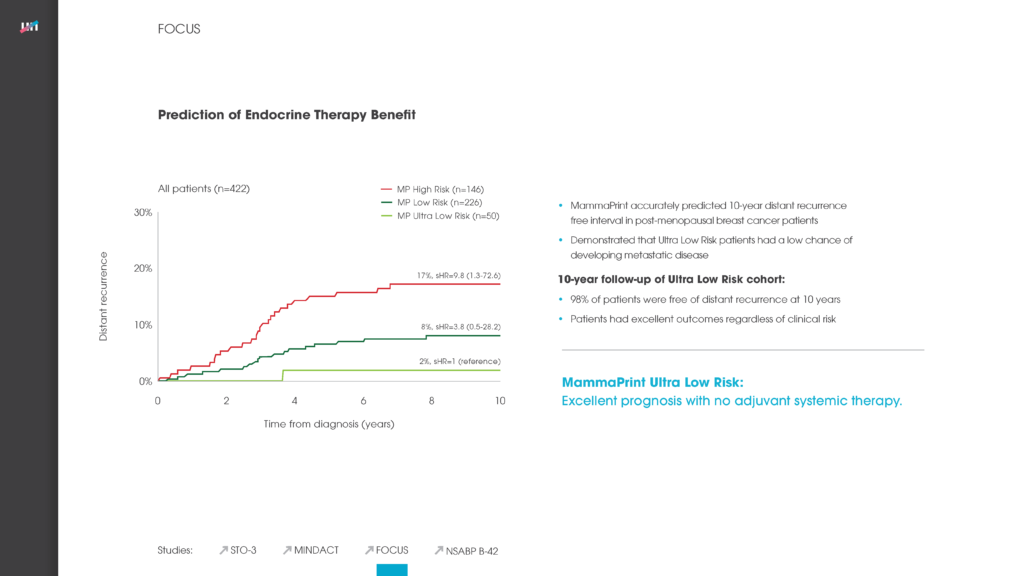
Among presentations by European researchers was a study concluding that the 70-gene MammaPrint recurrence test can accurately stratify a woman’s breast cancer risk of recurrence for up to 25 years after she is first diagnosed with the disease.
Another study at the EBCC conference updated findings from the ongoing Neoadjuvant Breast Registry Symphony Trial study. The data indicated that molecular subtyping using Agendia tests (MammaPrint and BluePrint®) is superior to ‘clinical subtyping’ by standard IHC/FISH pathology in predicting how a breast cancer patient will respond to neoadjuvant (pre-surgical) chemotherapy.
A separate podium presentation highlighted the accuracy and measurement concordance of the clinical biomarkers of ER, PR and Her2 thereby establishing the foundation for the methodology behind the prospective, 6,600 patient, multi-site, MINDACT study, a clinical trial comparing MammaPrint to standard clinical pathology factors. In a fourth presentation, a scientific poster from Dutch researchers demonstrated that physicians ordering the MammaPrint test are likely to follow its recommendations about whether or not adjuvant chemotherapy is advisable.
The study regarding MammaPrint’s ability to stratify a breast cancer patient’s recurrence risk for up to 25-years referenced above looked at two key measures regarding a set of 295 patients: distant metastasis-free survival (DMFS) and overall survival. The accuracy of MammaPrint results held true for both DMFS and overall survival. Median follow-up on these patients was 18.5 years – far longer than had previously been reported for a test of this kind.
The MINDACT podium presentation was by Fatima Cardoso, M.D. M.Sc., of Champalimaud Cancer Center in Lisbon, Portugal. She summarized research from 119 MINDACT trial sites in Belgium, Italy, Holland and Portugal. MINDACT is a multi-center, prospective, phase III randomized study that examines the role of MammaPrint in adding clinically useful information about tumor biology and assessment of recurrence risk, beyond information provided by standard clinical pathology such as tumor size and grade, patient age, number of involved lymph nodes, and ER/PR/HER2 status.
“These presentations featured exciting new research that shows the central role the Symphony tests can play in gaining a deeper, clinically useful understanding of the breast cancer biology and tumor subtypes at the molecular level,” said Neil Barth, M.D., a leading oncologist and Chief Medical Officer at Agendia.
MammaPrint, which is included in the guidelines of the European Society for Medical Oncology (ESMO), uncovers more clinically actionable biology, providing invaluable clinical information about an individual woman’s breast cancer and whether she is likely to experience a recurrence. The test delivers definitive High Risk or Low Risk information about the risk of recurrence, with no ambiguous “intermediate” results. It is the only assay providing risk recurrence information that is based on prospective trials including peer-reviewed patient outcome data (the RASTER study).
MammaPrint, which in its fresh tissue format has received five 510(k) FDA clearances, is performed as part of Agendia’s Symphony suite of breast cancer tests, which also includes BluePrint, the most widely available test providing molecular subtyping of individual breast cancers. MammaPrint has reimbursement arrangements collectively covering approximately 188 million lives in the U.S., including Medicare and contracts with a number of commercial payers, and is reimbursed in certain countries outside of the U.S. pursuant to regional coverage arrangements.
The 9th EBCC conference was held in Glasgow, U.K., March 19-21, 2014.
About Agendia:
Agendia is a leading molecular diagnostic company that develops and markets FFPE-based genomic diagnostic products, which help support physicians with their complex treatment decisions. Agendia’s breast cancer Symphony suite was developed using unbiased gene selection, analyzing the complete human genome. Symphony includes MammaPrint, as well as BluePrint®, a molecular subtyping assay, and TargetPrint®, an ER/PR/HER2 expression assay. MammaPrint is the only breast cancer recurrence assay with peer-reviewed, prospective outcome data. These tests can help physicians assess a patient’s individual risk for metastasis, which patients may benefit from chemo, hormonal, or combination therapy, and which patients may not require these treatments and can instead be treated with other, less arduous and less costly methods.
In addition to the Symphony suite of tests, Agendia has a pipeline of other genomic products in development. The company collaborates with pharmaceutical companies, leading cancer centers and academic groups to develop companion diagnostic tests in the area of oncology and is a critical partner in the ISPY-2 and the MINDACT trials. For more information, visit www.agendia.com.
Contact:
Liz Dowling (Medical & Consumer Media)
Dowling & Dennis Public Relations
415-388-2794
Liz@DowlingDennis.net
Len Hall (Financial Media)
Allen & Caron Inc. 949-474-4300