Agendia is disappointed with the recent update to the American Society of Clinical Oncology (ASCO) Clinical Practice Guidelines and with the decision to move forward without key research that we feel would have a material effect on the guidelines. We intend to introduce this information in the near future and trust that ASCO will consult Agendia in future guideline discussions.
In the first update to the guidelines since 2007, the authors concluded that the panel could not determine whether the assay identified a group of patients with either ER-positive or -negative, or node-positive or -negative, early-stage breast cancer for whom chemotherapy is either not required (prognosis) or not effective (prediction). We disagree with this conclusion.
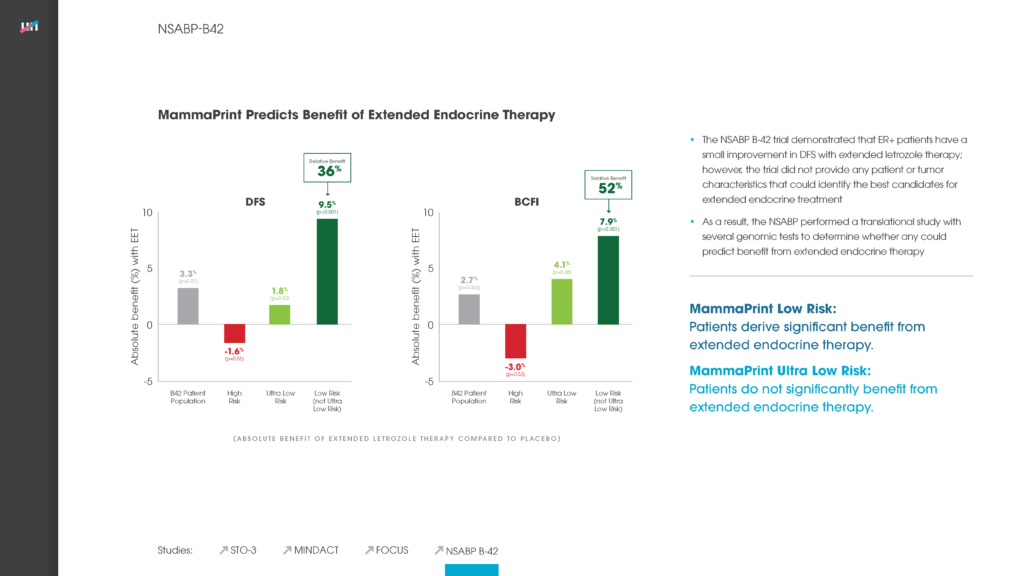
We feel that it’s critical that all relevant information is considered in developing guidelines. As part of this effort, Agendia is working to finalize new analyses that further validate the use of MammaPrint in this patient population. We look forward to sharing these findings as additional evidence to the added value the MammaPrint test provides physicians and their patients when considering adjuvant chemotherapy treatment.
***
For background, MammaPrint is acknowledged in recently-updated guidelines issued by the National Comprehensive Cancer Network® (NCCN®). These guidelines affirm that the MammaPrint test is clinically validated for prediction of prognosis of breast cancer in women of all ages and is not limited by estrogen receptor status. Furthermore, these guidelines cite published evidence underscoring MammaPrint’s position as the only breast cancer recurrence assay supported by peer-reviewed prospective outcome data. MammaPrint received FDA clearance in 2007 and is the first and only FDA cleared assay for use in risk assessment for women of all ages with breast cancer.