RASTER Five-Year Follow-Up Data Indicates Chemotherapy May Be Safely Withheld for Patients Deemed ‘Low Risk’ for Breast Cancer Recurrence Using Agendia’s MammaPrint Test, Data Presented at European Breast Cancer Conference
IRVINE, CA and AMSTERDAM, THE NETHERLANDS, March 22, 2012 – Agendia, an innovative molecular cancer diagnostics company, today announced that five-year follow-up data from the RASTER observational study indicates that its MammaPrint prognostic assay for breast cancer can help reduce adjuvant chemotherapy by 20 percent in patients deemed ‘Low Risk.’
From 2004 to 2006, the MicroarRAy PrognoSTics in Breast CancER study, known as RASTER, enrolled 427 women with primary breast carcinoma across 16 community-based hospitals in the Netherlands. The goal of the study was to assess feasibility of implementation and impact on treatment decisions of MammaPrint’s 70—gene prognosis signature. Initial study results, published in The Lancet Oncology in 2007, showed a 20 percent change in adjuvant treatment decisions after oncologists received MammaPrint diagnostic data.
“While there have been numerous studies that demonstrate the effectiveness of molecular diagnostic tests in identifying patients who are at risk for breast cancer recurrence, today marks the first time that data has been presented assessing the health outcomes of these individuals five years after receiving diagnostic results,” said Dr. Sabine Linn, Group Leader of the Netherlands Cancer Institute’s Division of Molecular Pathology and an investigator for the RASTER trial. “And the results are very significant.”
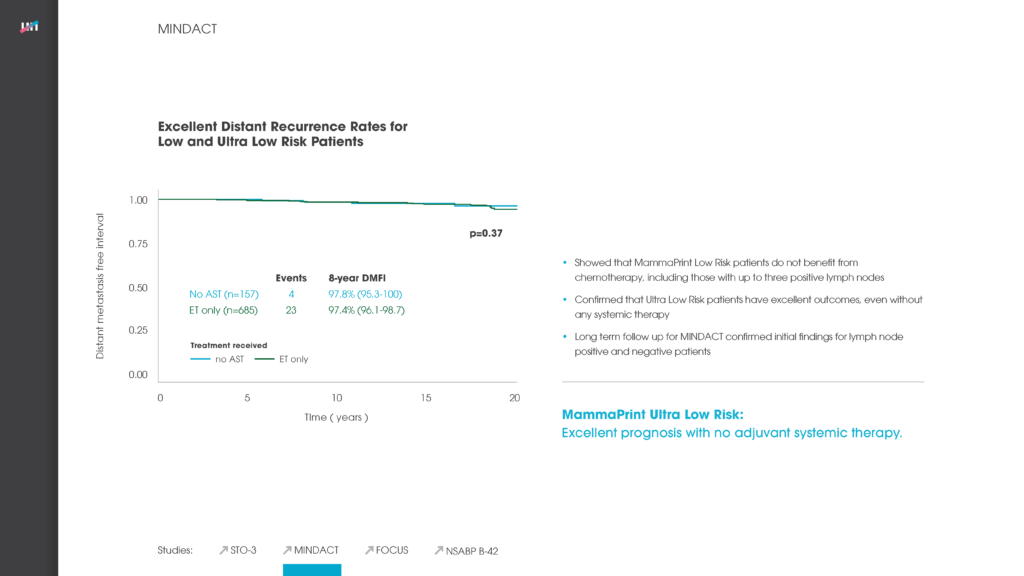
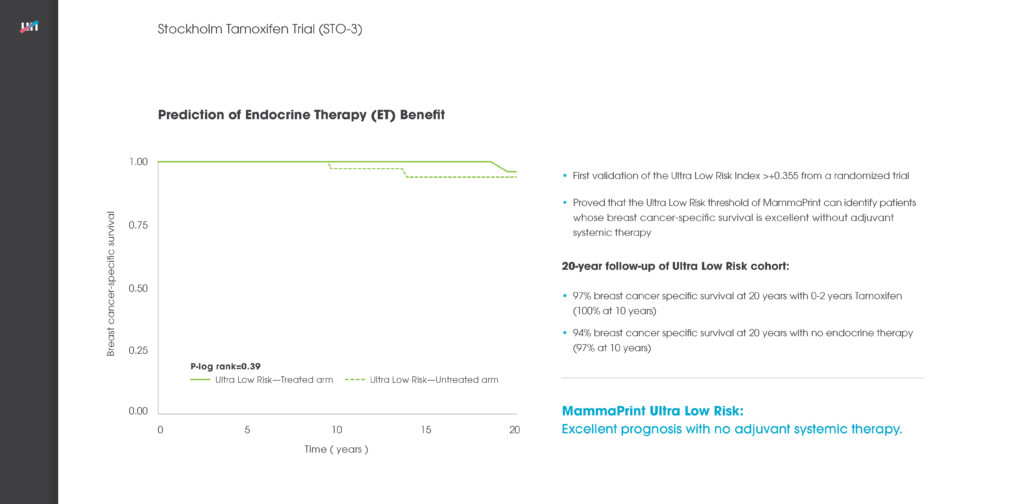
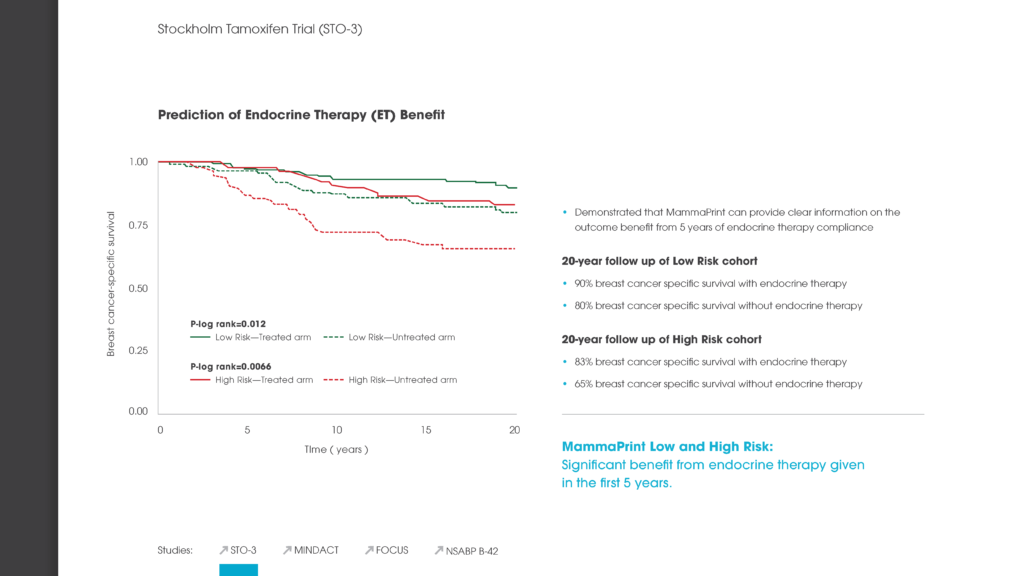
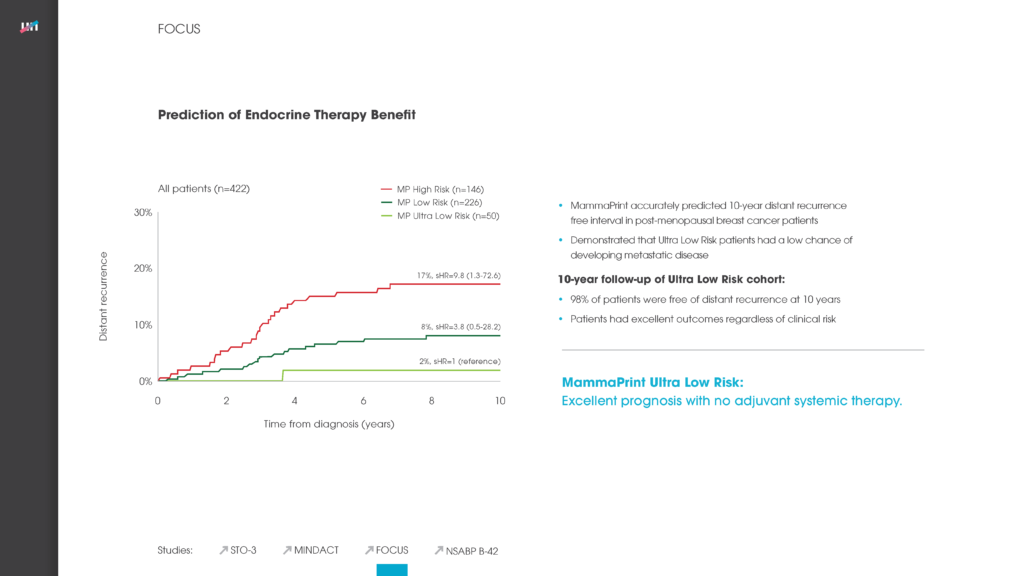
Specifically, nearly 51 percent of patients in the study were deemed ‘Low Risk’ for breast cancer recurrence via the MammaPrint assay, and the five-year distant metastasis-free survival (DMFS) rate for those patients was 96.1 percent. For patients deemed ‘High Risk’ for breast cancer recurrence, the five-year DMFS rate was 89.8 percent whereby the vast majority of these patients received chemotherapy. The data suggests that it is safe to withhold chemotherapy in low risk patient groups. The excellent five-year clinical outcome results for both low- and high-risk patients confirms the ability of MammaPrint to accurately stratify patients into appropriate treatment therapies.
“The five-year data from this prospective study clearly shows the promise of personalized medicine,” said David Macdonald, CEO of Agendia. “In an era of rising health care costs, MammaPrint informs physicians and patients as to the most effective course of treatment, reducing unnecessary costs and improving patient outcomes.”
About MammaPrint®
MammaPrint is the first and only breast cancer recurrence test cleared by the U.S. Food and Drug Administration (FDA). FDA clearance requires clinical and analytical validation and reporting systems to ensure patient safety issues are addressed. Highly accurate, MammaPrint identifies patients with early metastasis risk — patients who are likely to develop metastases within five years following surgery. Several authoritative studies have shown that chemotherapy particularly reduces early metastasis risk. In planning treatment, the MammaPrint test results provide doctors with a clear rationale to assess the benefit of chemotherapy in addition to other clinical information and pathology tests. All MammaPrint tests are conducted in Agendia’s CAP accredited, FDA compliant and CLIA certified service laboratories.
About Agendia:
Agendia is a leading molecular diagnostic company that develops and markets genomic-based diagnostic products that improve the quality of life for cancer patients and simplifies complex treatment decisions for their physicians. Agendia’s Symphony™ suite of breast cancer products is based on the analysis of hundreds of genes in a patient’s breast tumor biology and provides unprecedented clinical insight to address complex treatment decisions. Symphony includes MammaPrint, the first and only FDA-cleared IVDMIA breast cancer recurrence assay, as well as BluePrint, a molecular subtyping assay, TargetPrint®, an ER/PR/HER2 expression assay, and TheraPrint®, a therapy selection assay. Together, these tests help physicians determine a patient’s individual risk for metastasis, which patients will benefit from chemo, hormonal, or combination therapy, and which patients do not require these treatments and can instead be treated with other less arduous and less costly methods.
In addition to the Symphony suite of tests, Agendia has a rich pipeline of genomic products in development. The company collaborates with pharmaceutical companies, leading cancer centers and academic groups to develop companion diagnostic tests in the area of oncology and is a critical partner in the ISPY-2 and MINDACT trials.
Agendia was founded in 2003 as a spin-off of the Netherlands Cancer Institute and is based in Irvine, California, United States, and Amsterdam, the Netherlands. For more information, please visit www.agendia.com.
For further information, please contact:
Ricochet Public Relations
Todd Aydelotte
Tel: +1 212 679 3300
E-mail: todd@ricochetpr.com