PUBLICATION:
Expert Rev Pharmacoecon Outcomes Res. 2014 Dec;14(6):889-99. doi: 10.1586/14737167.2014.957185. Epub 2014 Sep 12.
AUTHORS:
Seguí M.Á., Crespo C., Cortés J., Lluch A., Brosa M., Becerra V., Chiavenna S.M., Gracia A.
SUMMARY:
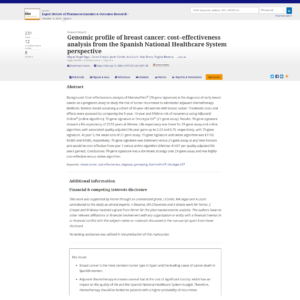
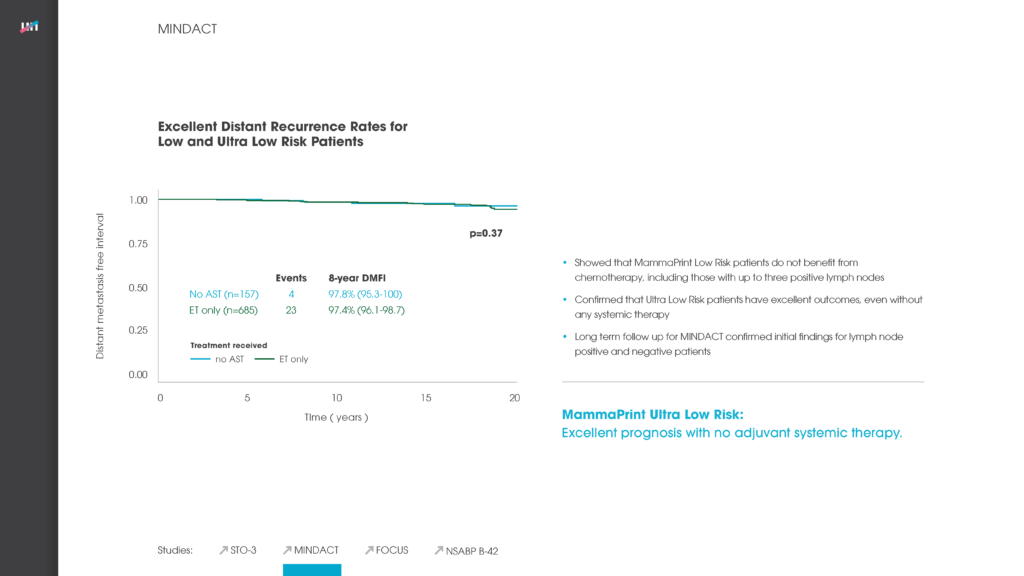
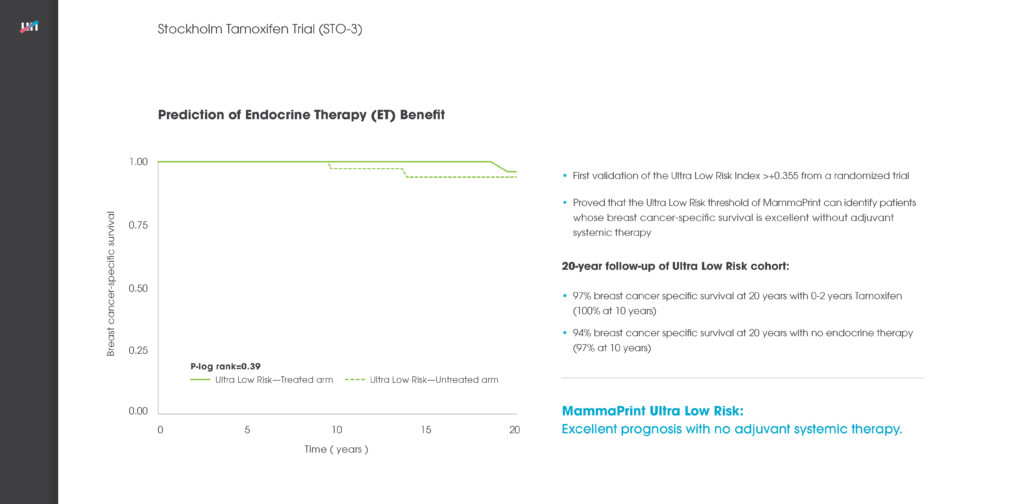
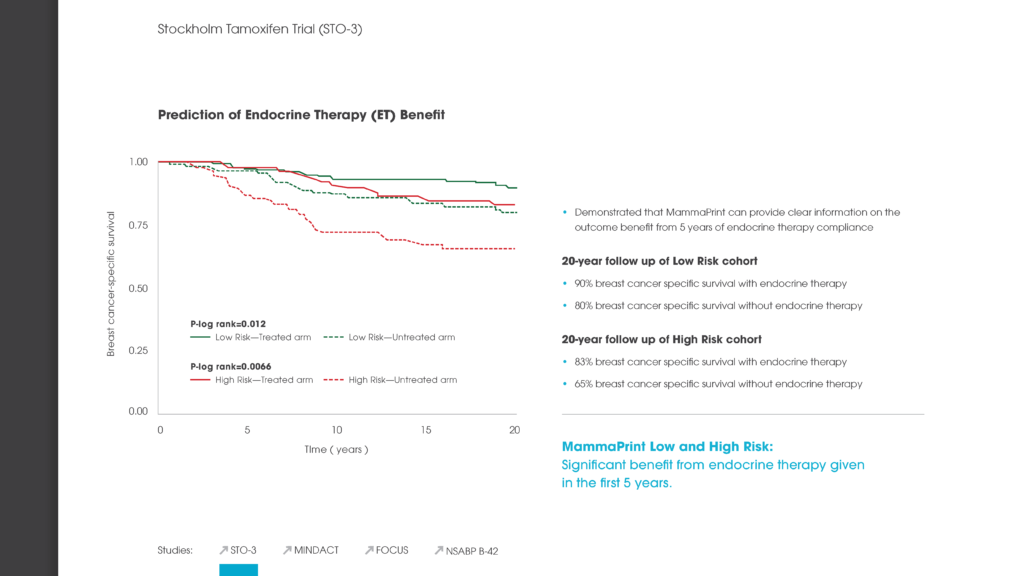
A 70-gene signature showed a life expectancy of 23.55 years at lifetime. Life expectancy was lower for 21-gene assay and online algorithm, with associated quality-adjusted life year gains up to 0.23 and 0.75, respectively, with 70-gene signature. At year 5, the mean cost of 21-gene assay, 70-gene signature and online algorithm was €7100, €6380 and €4580.