CHICAGO – Molecular subtyping can help predict outcomes and chemotherapy benefit in patients with colorectal cancer, according to a presentation at the annual conference of the American Society of Clinical Oncology (ASCO). This scientific poster was one chosen for discussion at the ASCO conference due to the significance of the scientific discovery for treating colorectal cancer patients.
In the multi-site study, researchers identified three major molecular subtypes of colorectal cancer and determined that these subtypes are significantly associated with prognosis and response. They also determined the subtypes are linked to deficient mismatch repair, mutation frequency, epithelial-to-mesenchymal transition, and different activated pathways.
All this information indicates there is a difference in underlying biology. Therefore the new diagnostic classification provides clinically valuable information about an individual patient’s prognosis and how likely it is that the patient would benefit from adjuvant (post-surgical) chemotherapy. The molecular subtype classification was validated in 543 Stage II and Stage III patients and in stage I to IV patients in the widely recognized TCGA (The Cancer Genome Atlas) dataset.
“Different patients have different responses to treatment for colorectal cancer because of the underlying biology of the tumor. Having this new information about molecular subtypes provides valuable information regarding prognosis and treatment,” said poster co-author Scott Kopetz, M.D., Ph.D., FACP, a medical oncologist at The University of Texas MD Anderson Cancer Center. “We now know that, along with clinical-pathological risk factors, the subtype is clinically relevant in the treatment decision process.”
The research was presented by primary author Ramon Salazar, M.D. Ph.D, of the Catalan Institute of Oncology, in Barcelona, Spain, along with Dr. Kopetz. Other co-authors of the study were from the Netherlands, Germany, and Austria.
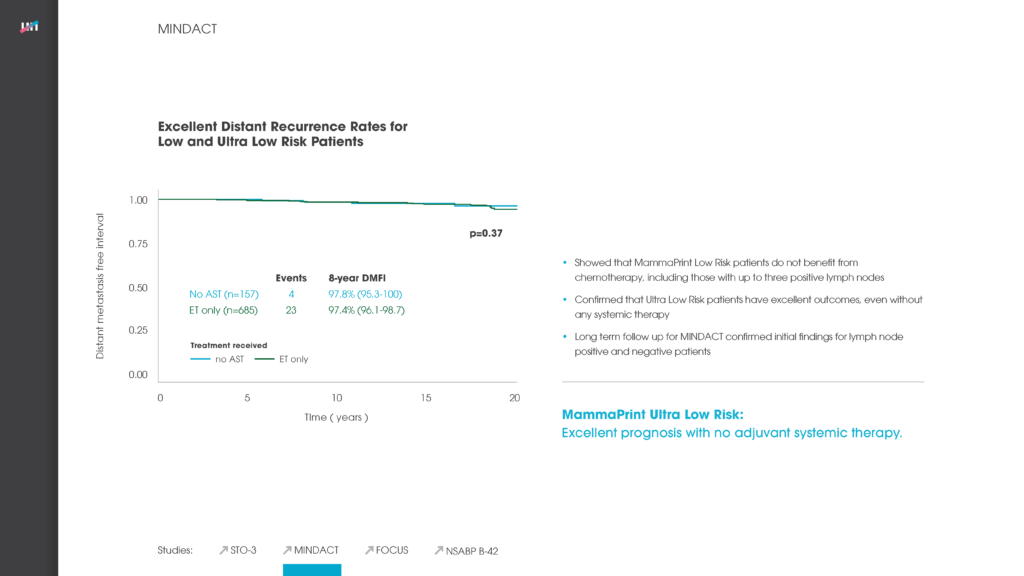
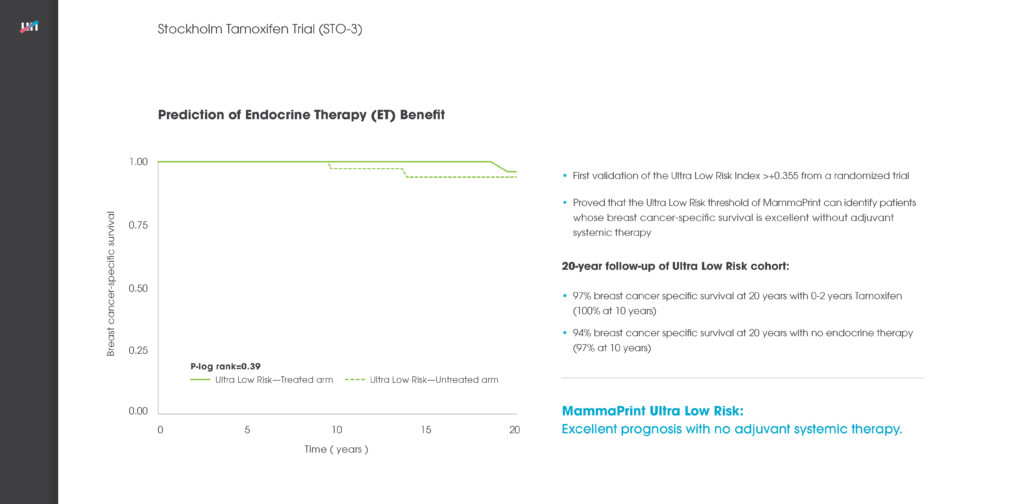
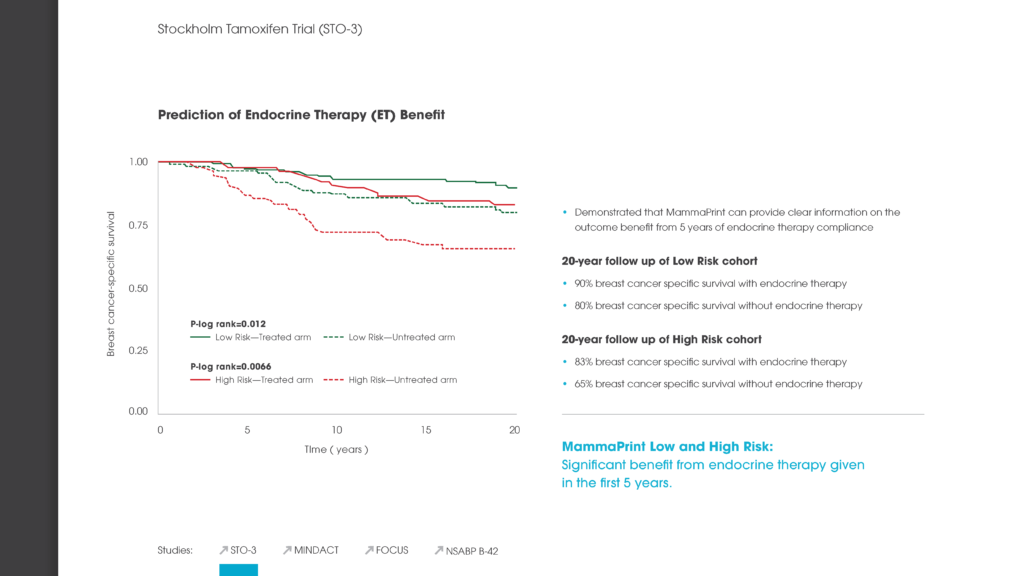
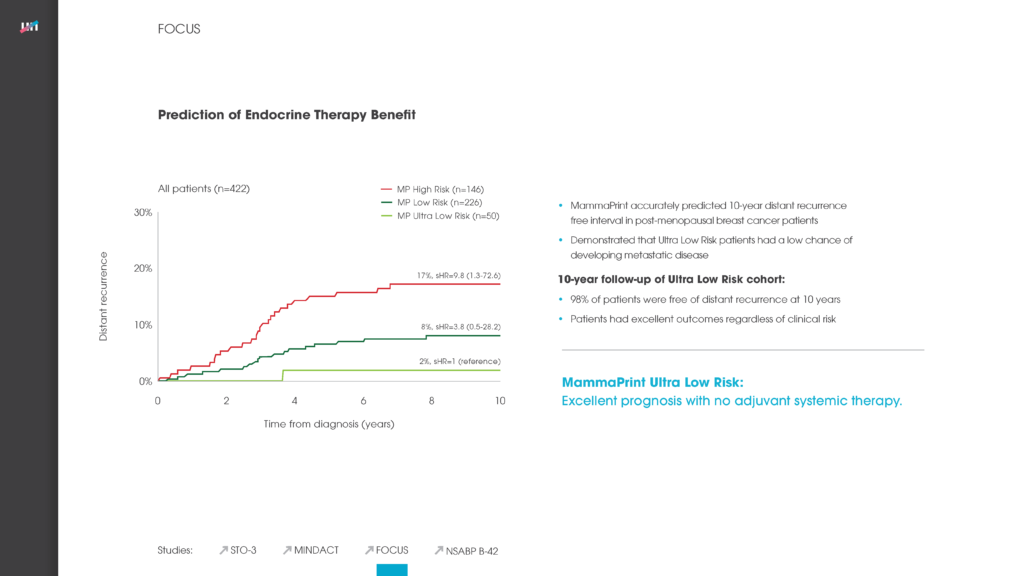
Another poster at the ASCO meeting reported on the new independent validation study of the genomic classifier ColoPrint, made by Agendia, which predicts outcome and chemotherapy benefit in Stage II and III colon cancer patients. The study concluded that ColoPrint significantly improves prognostic accuracy in Stage II and Stage III colon cancer patients — thereby supporting physicians’ decisions on which patients would most benefit from chemotherapy and which patients would not.
Three posters regarding breast cancer reported on:
- The effectiveness of MammaPrint® and BluePrint® in identifying molecular subtypes of breast tumors as a predictor for tumor response to targeted chemotherapies
- The MammaPrint MINT study assessing the ability of molecular profiling and traditional pathologic and clinical prognostic factors to predict patient response to neo-adjuvant chemotherapy.
- Genomic profiling of breast cancer in African American women.
The ASCO meeting was held in Chicago May 31-June 4, 2013.
About Agendia
Agendia is a leading molecular diagnostic company that develops and markets FFPE-based genomic diagnostic products. Agendia’s breast cancer Symphony™ suite was developed by analyzing the complete human genome, ensuring 100% definitive results for breast cancer patients. Together, these tests help physicians determine a patient’s individual risk for metastasis, which patients will benefit from chemo, hormonal, or combination therapy, and which patients do not require these treatments and can instead be treated with other less arduous and less costly methods.
In addition to the ColoPrint genomic classifier and Symphony suite of tests, Agendia has a rich pipeline of genomic products in development. The Company collaborates with pharmaceutical companies, leading cancer centers and academic groups to develop companion diagnostic tests in the area of oncology and is a critical partner in the ISPY-2 and MINDACT trials. For more information, please visit www.agendia.com
Liz Dowling
(415) 388-2794
Dowling & Dennis Public Relations
E-mail: Liz@DowlingDennis.net