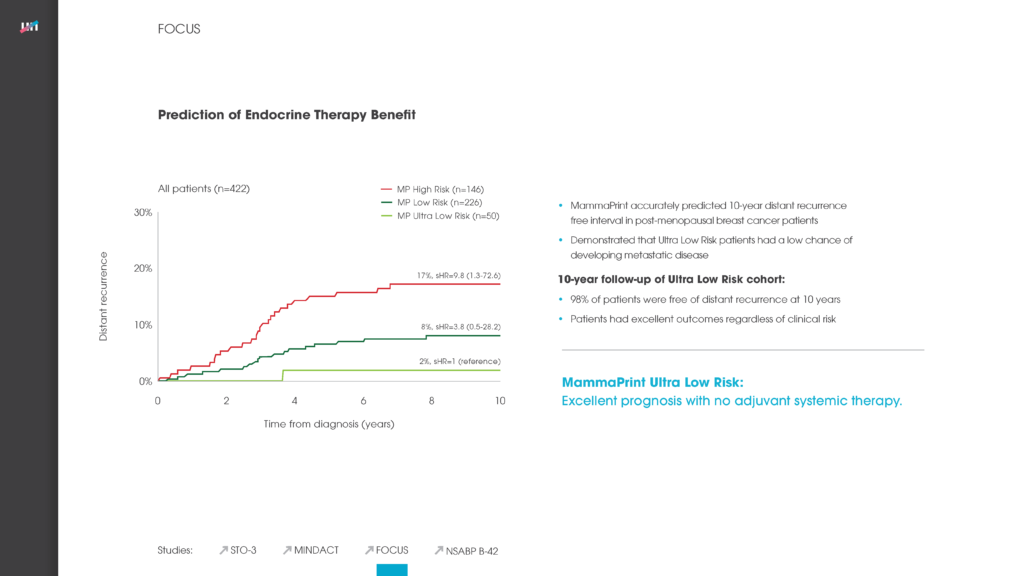
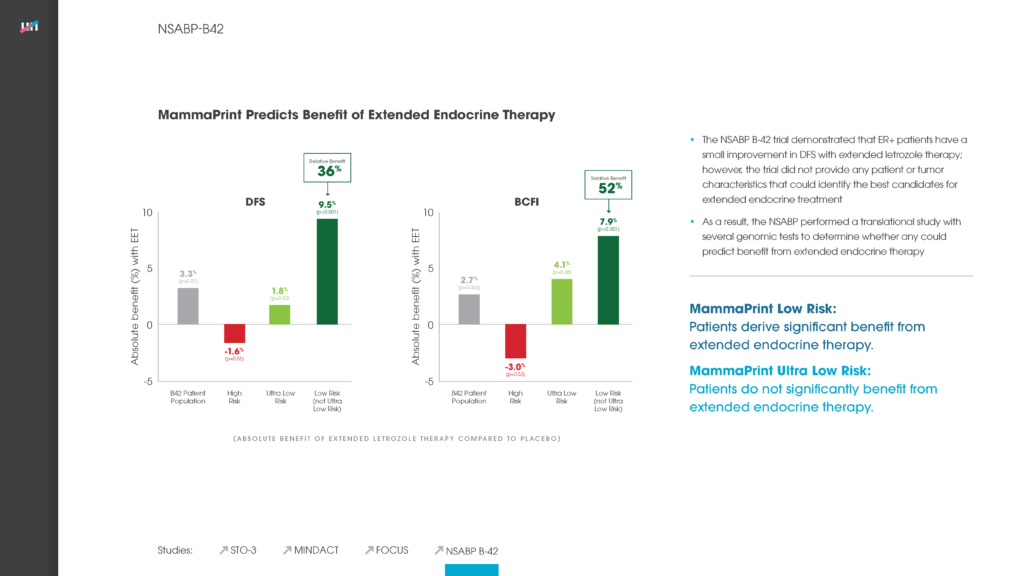
PUBLICATION:
Breast Cancer Res Treat. 2010 Apr;120(3):655-61. doi: 10.1007/s10549-010-0814-2.
AUTHORS:
Knauer M., Mook S., Rutgers E.J., Bender R.A., Hauptmann M., van de Vijver M.J., Koornstra R.H., Bueno-de-Mesquita J.M., Linn S.C., van ‘t Veer L.J.
ABSTRACT:
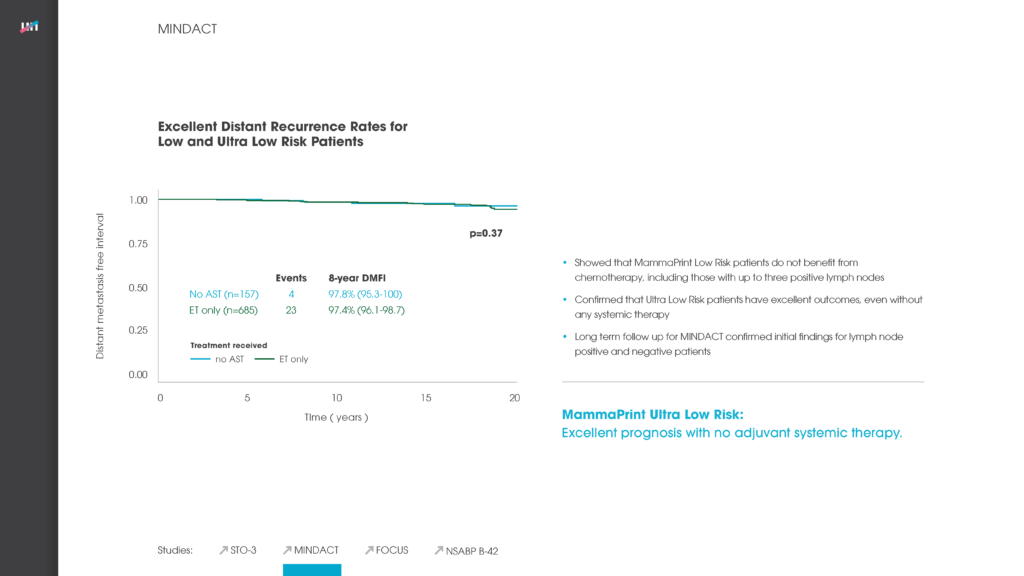
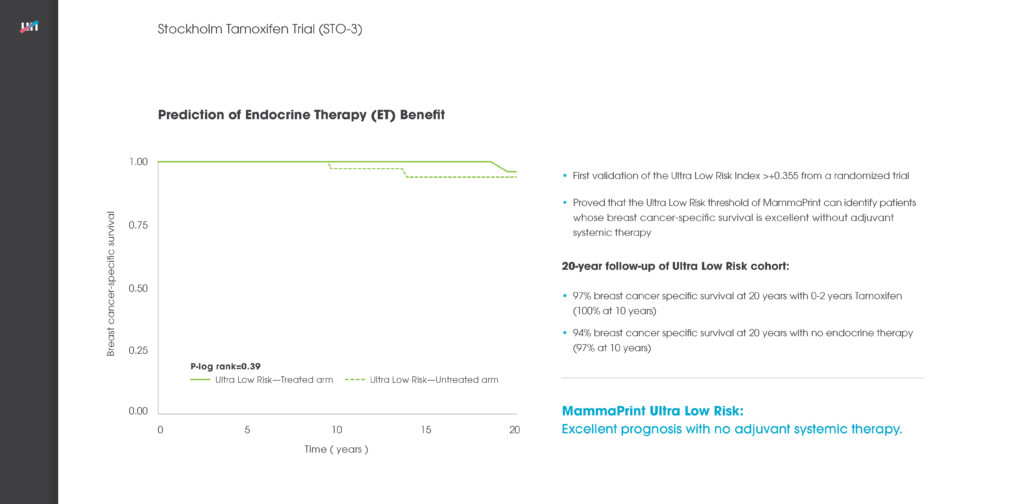
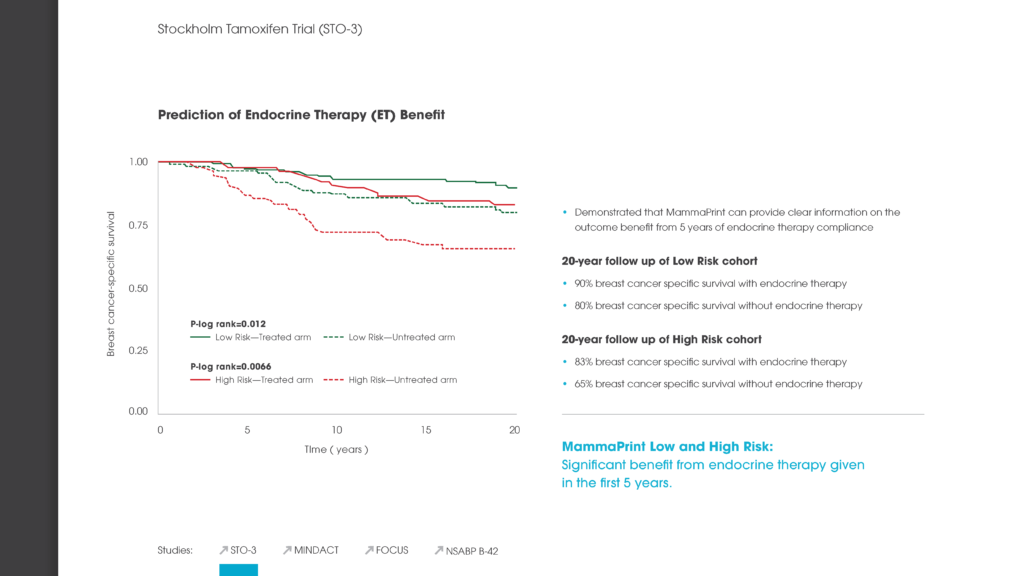
Multigene assays have been developed and validated to determine the prognosis of breast cancer. In this study, we assessed the additional predictive value of the 70-gene MammaPrint signature for chemotherapy (CT) benefit in addition to endocrine therapy (ET) from pooled study series. For 541 patients who received either ET (n = 315) or ET + CT (n = 226), breast cancer-specific survival (BCSS) and distant disease-free survival (DDFS) at 5 years were assessed separately for the 70-gene high and low risk groups. The 70-gene signature classified 252 patients (47%) as low risk and 289 (53%) as high risk. Within the 70-gene low risk group, BCSS was 97% for the ET group and 99% for the ET + CT group at 5 years with a non-significant univariate hazard ratio (HR) of 0.58 (95% CI 0.07-4.98; P = 0.62). In the 70-gene high risk group, BCSS was 81% (ET group) and 94% (ET + CT group) at 5 years with a significant HR of 0.21 (95% CI 0.07-0.59; P < 0.01). DDFS was 93% (ET) versus 99% (ET + CT), respectively, in the 70-gene low risk group, HR 0.26 (95% CI 0.03-2.02; P = 0.20). In the high risk group DDFS was 76 versus 88%, HR of 0.35 (95% CI 0.17-0.71; P < 0.01). Results were similar in multivariate analysis, showing significant survival benefit by adding CT in the 70-gene high risk group. A significant and clinically meaningful benefit was observed by adding chemotherapy to endocrine treatment in 70-gene high risk patients. This benefit was not significant in low risk patients, who were at such low risk for recurrence and cancer-related death, that adding CT does not appear to be clinically meaningful.
Read more: Predictive Value of MammaPrint_Knauer M et al_BCRT 2010